Cleanrooms Medical Device
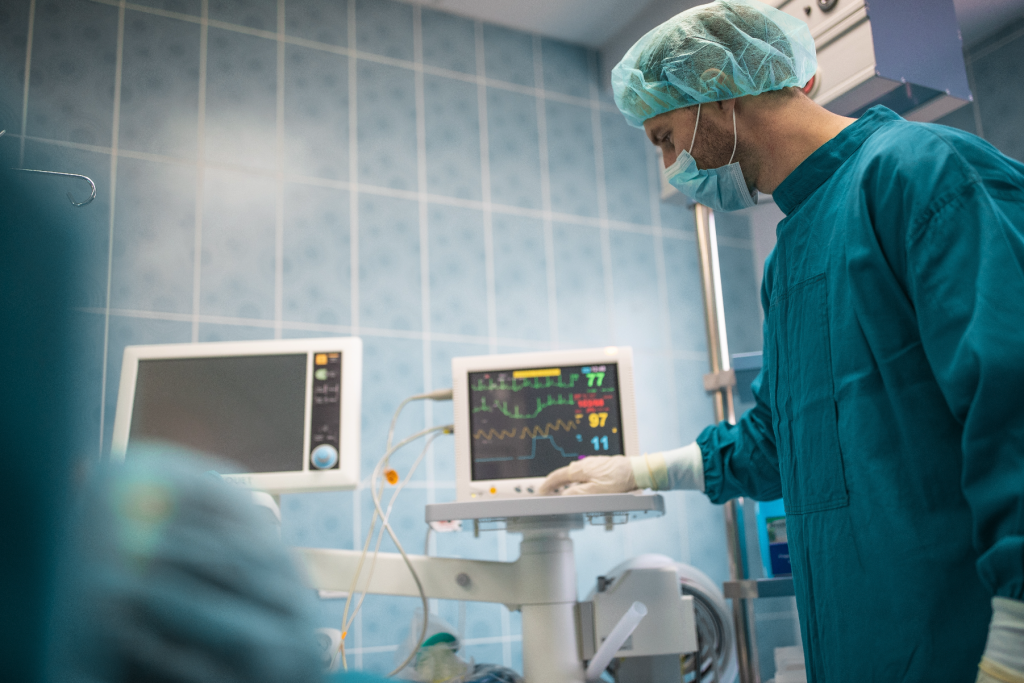
In the realm of medical devices, precision is not just a preference; it’s an absolute necessity. The manufacturing of medical devices demands unparalleled sterility, reliability, and adherence to strict quality standards. Cleanroom Design emerges as a cornerstone in this endeavour, specialising in the construction of cleanrooms tailored specifically for the medical device industry. In this […]
Cleanroom Pharma

In the pharmaceutical industry, precision is paramount. The production of life-saving medications, vaccines, and pharmaceutical products demands not only scientific excellence but also unwavering sterility and quality. This is where Cleanroom Design steps onto the stage, playing a pivotal role in constructing cleanrooms tailored specifically for the pharmaceutical sector. In this article, we explore the […]
The Building Blocks of Cleanroom Compliance: Steering Regulatory Considerations through Project Lifecycles

In the meticulously controlled world of cleanrooms, every design element and construction phase is a testament to precision and compliance. Whether serving the pharmaceutical, biotechnological, or electronic sectors, cleanrooms are the bedrock of controlled, contamination-free environments crucial for product reliability and safety. In the UK, steering a cleanroom project from blueprint to operational readiness demands […]
New Annex 1 Requirements

The newly revised Annex 1 requirements, set to come into effect on August 25, 2023, signify a substantial step forward in the regulation of cleanroom environments, particularly in the manufacture of sterile medicinal products. Here’s a detailed look at the pivotal updates in the new Annex 1 requirements: Contamination Control Strategy (CCS): One of the […]
New Annex 1 Requirements and Practical Changes Within Cleanrooms
The amendments stipulated in the new Annex 1 requirements will manifest in several practical changes within cleanrooms, especially those involved in the manufacture of sterile medicinal products. Here are some real-world examples of how these changes might take shape: Implementation of a Contamination Control Strategy (CCS): Cleanrooms will now have a structured Contamination Control Strategy […]
